Description
(USP 34)
- Drug Substance General Information (ICH 3.2.S.1)
1.1. Name(ICH 3.2.S.1.1)
International non-Proprietary Name: Alendronate sodium (Brand Name: Fosamax)
Compendial Name: Alendronate sodium
Chemical Name: (4-amino-1-hydroxybutylidene)bis- phosphonicacid,monosodium salt, trihydrate; Sodium trihydrogen (4-amino-1-hydroxybutylidene)diphosphonate, trihydrate
Arasto’s code: ALE
CAS Registry Number: [121268-17-5]
- Drug Substance General Information (ICH 3.2.S.1)
1.2. Structure (ICH 3.2.S.1.2)

Empirical Formula: C4H12NNaO7P2 . 3H2O
Molecular Weight: 325.12
- Drug Substance General Information (ICH 3.2.S.1)
1.3. General Properties (ICH 3.2.S.1.3)
Alendronate sodium is a white to off-white crystalline substance. It is orally administered for the prevention and the treatment of osteoporosis in postmenopausal women, treatment to increase bone mass in men with osteoporosis and the treatment of Paget’s disease of bone. Alendronate sodium is slightly soluble in neutral cold water and soluble in hot water and insoluble most organic solvents. Alendronic acid has pKa values of 0.8, 2.2, 6.3 and 12.2 and very soluble in water at very low pH due to the formation of alendronicacid (Ezra, A., Colombo, G. Adv. Drug Deliv.Rev. 2000, 42, 175). Its log P has been reported to be -3.2 (http://pharmacycode.com/ Alendronate Sodium.html). Alendronate sodium is stable in acid and in base (see Stability Studies). ItsLD50 in female rats and mice were 552 mg/kg and 996 mg/kg respectively (http://www.labriva.com/media/monographies/RIVA-Alendronate40mg70mgP-08-24.pdf).
The determination of purity and assay of APIs require comparison of the product with their respective Reference Standards (RS) and Related Compounds (RC or known impurities). Accordingly, ICH regulations on the purity and assay of reference standard and related compounds are clearly defined and must be followed by drug substance and drug product manufacturers.
According to ICH Q7, 11.1 there are 3 types of standards. This is summarized in the following chart and discussed in detail below.

The impurities provided in the following table represent Secondary Reference Standards (SRS) that are prepared in-house by synthesis or by isolation. Each SRS has undergone extensive characterization ( IR, UV, 1HNMR, 13CNMR. Mass Spec) and determination of its purity and assay (HPLC). For specification of the SRS of those products that have a monograph, the SRS is compared with a pharmacopoeia Primary Reference Standard (UV, HPLC retention time). For specification of those products that do not have a monograph (known as House Primary Standard), we compare their UV ε or ג/max , IR major absorptions, 1HNMR d (ppm) , 13 CNMR d (ppm) or HPLC retention time with values reported in the chemical literature for these compounds.

- Primary and Secondary Reference Standard (ICH 3.2.S.5)
5.1. Active Pharmaceutical Ingredient
Primary Reference Standard for Alendronate sodium is available from United States Pharmacopoeia. We will use a Secondary Reference Standard (previously referred to as Working Standard) for direct control of all batches of Alendronate sodium.
As per ICH (Q7, 11.1) and ICH (Q6, 2.11, 3.2, 3.3)the Secondary Reference Standards, which include the API and its Related Compounds, must be examined for their proof of structure (characterization), assay and purity and specification (identification by comparison). Furthermore, ICH Guideline on the Preparation of Common Technical Document (Q4M) requires that the data obtained from characterization, assay and purity and specification must be included in section 3.2.S.3.2 for Related Compounds (already discussed in that section) and section 3.2.S.5 of the DMF for the API. To this end, the Secondary Reference Standards of the API Alendronate sodium has undergone extensive characterisation (UV, IR, 1 H NMR, 13C NMR, and Mass Spec) to assure its structure, assay and purity (HPLC and/or titration) and specification (comparison of its HPLC retention time and UV ג/max with USP Primary Reference Standard.
The Secondary Reference Standard for Alendronate sodium was produced from a released batch of Alendronate sodium by subjecting it to an additional crystallization from the final solvent system used in the production of the API to avoid the possibility of other polymorph formation.
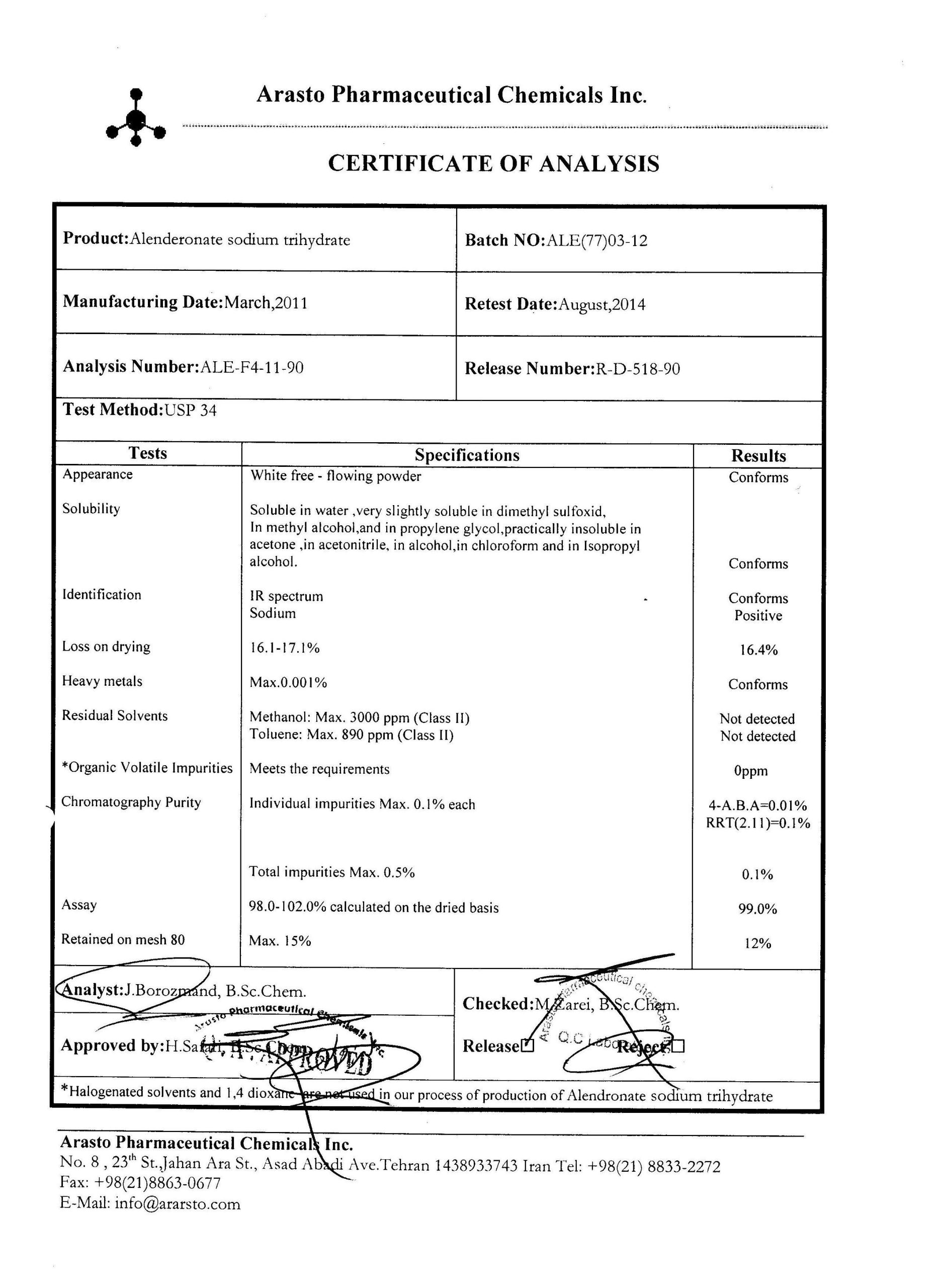
Certificate of analysis of a typical batch with USP method