Description
Description
(USP 44)
Description
- Drug Substance General Information (ICH 3.2.S.1)
1.1. Nomenclature (ICH 3.2.S.1.1)
International non-proprietary name: Oxaliplatin (Brand Name: Eloxatin)
Compendial name: Oxaliplatin
Chemical name: [SP-4-2-(1R-trans)]-(1, 2-Cyclohexanediamine-N,N′)[ethanedioato(2-)-O,O’] platinum.
Arasto’s code: OXA
CAS Registry Number: [61825-94-3].
Official Pharmacopoeia Monograph: USP 44
Drug Substance General Information (ICH 3.2.S.1)
1.2. Structure (ICH 3.2.S.1.2)

Empirical Formula: C8H14N2O4Pt
Molecular Weight: 397.29 g/mol
- Drug Substance General Information (ICH 3.2.S.1)
- General Properties (ICH 3.2.S.1.3)
Oxaliplatin is a white to off-white powder and it is slightly soluble in water at 6 mg/mL, very slightly soluble in methanol, and practically insoluble in ethanol and acetone at 25°C. Oxaliplatin melting point is over 245°C and about 260°C. Decomposition temperature is not established. 5 mg per ml of Oxaliplatin in distilled water is clear and has Specific rotation of between +74.58 and +78.08 at 20°C. A 5mg/ml solution of Oxaliplatin in water has a pH of 4.0-7.0 at 25°C. (http://www.sagentpharma.com/wp-content/uploads/2014/12/Oxaliplatin_SDS.pdf). Oxaliplatin (1-OHP; L-OHP) is an anti-cancer (“antineoplastic” or “cytotoxic”) chemotherapy drug. Oxaliplatin is classified as an “alkylating agent” and it is used to treat colon or rectal cancer that has spread (metastasized). It is often given in combination with other anticancer drugs (fluorouracil and leucovorin).
The determination of purity and assay of APIs require comparison of the product with their respective Reference Standards (RS) and Related Compounds (RC or known impurities). Accordingly, ICH regulations on the purity and assay of reference standard and related compounds are clearly defined and must be followed by drug substance and drug product manufacturers.
According to ICH Q7, 11.1 there are 3 types of standards. This is summarized in the following chart and discussed in detail below.
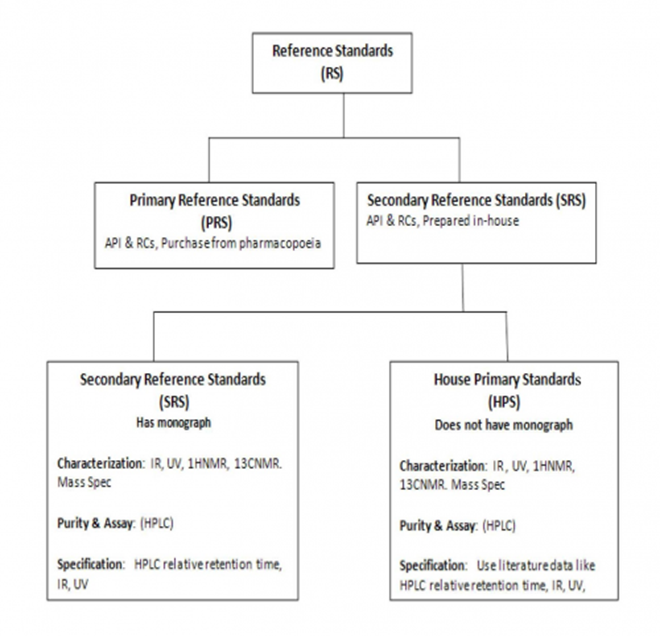
The impurities provided in the following table represent Secondary Reference Standards (SRS) that are prepared in-house by synthesis or by isolation. Each SRS has undergone extensive characterization (IR, UV, 1HNMR, 13CNMR. Mass Spec) and determination of its purity and assay (HPLC). For specification of the SRS of those products that have a monograph, the SRS is compared with a pharmacopoeia Primary Reference Standard (UV, HPLC retention time). For specification of those products that do not have a monograph (known as House Primary Standard), we compare their UV ε or ג /max , IR major absorptions, 1HNMR d (ppm) , 13 CNMR d (ppm) or HPLC retention time with values reported in the chemical literature for these compounds.
Oxaliplatin Related Compounds
| Structure | Chemical Name | USP Code | Arasto Code |
| oxalic acid dihydrate | RCA | RCA | |
| [SP– 4-2-(1R-trans)]-(1,2-cyclohexanediamine-N,N’) dinitratoplatinum(II) | RCB | RCB | |
| [1R-trans-(1,2- cyclohexanediamine-N,N’)]-trans-dihydroxido-[oxalato(2-) – O,O’]platinum(IV) | RCC | RCC | |
| cis– [(1S,2S)-1,2-cyclohexanediamine-N,N’][oxalato(2-)-O,O’]platinum | RCD | RCD | |
| (SP-4-2)-di-μ-oxobis[(1R,2R)-cyclohexane-1,2-diamine-N,N′]diplatinum | Diaquodiaminocyclo-hexaneplatinum dimer | RCE |
5. Primary and Secondary Reference Standard (ICH 3.2.S.5)
5.1. Active Pharmaceutical Ingredient
Primary Reference Standard for Oxaliplatin is available from United States Pharmacopoeia. We will use a Secondary Reference Standard (previously referred to as Working Standard) for direct control of all batches of Oxaliplatin.
As per ICH (Q7, 11.1) and ICH (Q6, 2.11, 3.2, 3.3) House Primary Standards, which include the API and its Related Compounds, must be examined for their proof of structure (characterization), assay and purity and specification (identification by comparison). Furthermore, ICH Guideline on the Preparation of Common Technical Document (Q4M) requires that the data obtained from characterization, assay and purity and specification must be included in section 3.2.S.3.2 for Related Compounds (already discussed in that section) and section 3.2.S.5 of the DMF for the API. To this end, the House Primary Standard of the API Oxaliplatin has undergone extensive characterization (UV, IR, 1 H NMR, 13C NMR, Mass Spec) to assure its structure, assay and purity (HPLC and/or titration) and specification.
The Secondary Reference Standard for Oxaliplatin was produced from a released batch of Oxaliplatin by subjecting it to an additional crystallization from the final solvent system used in the production of the API to avoid the possibility of other polymorph formation.
SPECIFICATION OF ANALYSIS
| Product: Oxaliplatin | CAS No.: 61825-94-3 | Spec. No.: APC-QC-SPEC-360-00 | ||
| Issue Date: Apr, 2023 | Valid up to: Apr, 2024 | Reference: USP44 | ||
| Tests | Specifications | |||
| Description | White to off- white crystalline powder. | |||
| Solubility | Slightly soluble in water, very Slightly soluble in methanol, practically insoluble in alcohol. | |||
| Identification | A: Infrared Absorption B: The retention of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay. | |||
| Specific Rotation | Between +74.5° and +78.0°, measured at 20° (5mg/ml in water) | |||
| Loss on drying | NMT 0.5% (at 100°-105° for 2 h) | |||
| Acidity | The solution is colorless, and NMT 0.6 ml of 0.01 M Sodium hydroxide is required to change the color to pink. | |||
| Content of platinum | 48.1%–50.1% of the oxaliplatin taken on the dried basis | |||
| Limit of silver | NMT 5ppm | |||
| Organic impurities, procedure 1 | Oxalic acid: NMT 0.1% | |||
| Organic impurities, procedure 2 | Oxaliplatin related compound C: NMT 0.1% (SP-4-2)-diaqua [(1R,2R)-cyclohexane-1,2-diamine-N,N’] platinum: NMT 0.1% Diaquodiaminocyclohexaneplatinum dimer: NMT 0.1% Any individual unspecified impurity: NMT 0.10% Total impurities: NMT 0.30% | |||
| Organic impurities, procedure 3 | Oxaliplatin related compound D: NMT 0.1% | |||
| Residual solvent | Ethanol: NMT 5000 ppm (Class III) Diethyl ether: NMT 5000 ppm (Class III) | |||
| Assay | 98.0% to 102.0% (on the dried basis) | |||
| Bacterial Endotoxins test | NMT 1.0 USP Endotoxin Unit per mg of Oxaliplatin. | |||
| Microbial enumeration tests and Tests for specified microorganisms | The total aerobic microbial count does not exceed 20 cfu/g and the total combined molds and yeast count does not exceed 5 cfu/g. | |||
| Prepared by: M. Shahbazi, B.Sc.Chem. | Checked by: A. Forghani, B.Sc.Chem. | |||
| Approved by: F. Javadizadeh, M.Sc.Chem. | ||||
| Storage: Preserve in tight containers, and store at a temperature below 30°. | ||||